|
12/4/2023 0 Comments Atomic size trend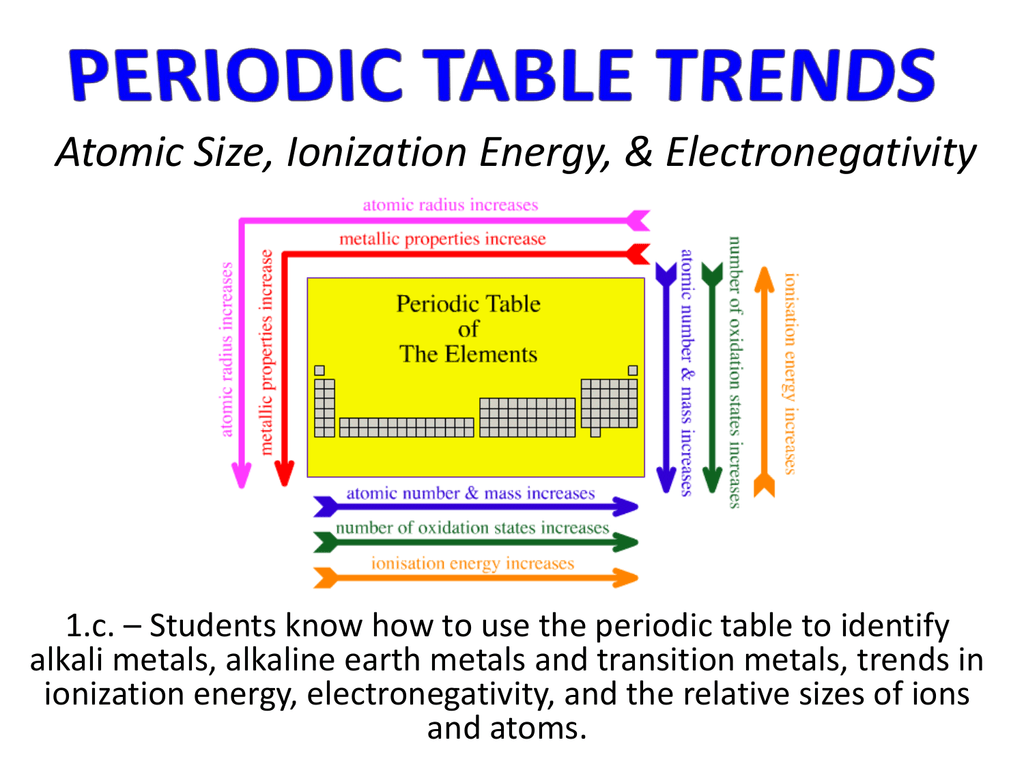
Any opinions expressed on this website are entirely mine, and do not necessarily reflect the views of any of my employers. All text and images on this website not specifically attributed to another source were created by me and I reserve all rights as to their use. Jeff Cruzan is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License. Of sodium (Na) or rubidium (Rb), which has the greater first ionization energy? Solution Of phosphorus (P) or iodine (I), which has the greater first ionization energy? Solution Why does fluorine (F) have a higher first ionization energy than iodine (I)? Solution Which has the least attraction for electrons in a chemical bond? – O, Cl, F, N Solution Hypothesize about why 90Sr might cause bone cancers, leukemias and other cancers in children exposed to such nuclear accidents. One of the highly-possible products of fallout from nuclear explosions or nuclear power plants is 90Sr, a radioactive isotope of strontium (Sr). :max_bytes(150000):strip_icc()/PeriodicTable_AtomSizes-56a131193df78cf772684720.png)
Which atom of each pair has the largest atomic radius? (a) Al | B (b) Na | Al (c) Mg | Ca (d) O | F (e) Br | Cl (f) S | O Solution Rank these elements in order of increasing electronegativity: O, S, Ne, Al Solution Rank these elements in order of increasing atomic radius: C, Al, O, K Solution As you move up and to the right, from francium (Fr) to fluorine (F), electronegativity increases. For example, Xe and strongly electronegative compounds like fluorine (F) and oxygen (O) to form compounds like XeF, XeF 6 XeO 3F 2īelow is a simple periodic table showing the gross trend – without any exceptions – of electronegativity. They do react to form some compounds, however. These electronegativities can't be scaled directly with those of, say, the halogens, because large noble gases don't react in the same ways to make similar compounds. Noble gasesįinally, what's up with the electronegativities of krypton (Kr), xenon (Xe) and radon (Rn) ? Tungsten, on the other hand, is not such an exception, so a borrowed 5 th 3d electron is energetically favorable, thus its relatively high electronegativity. For example, the lowest-energy electron configuration of Cr is They take an s electron from an s orbital in order to half-fill the d-orbital below it. Recall that the electron configurations of these elements are also exceptions. We can make a similar argument as we move down the 6B group from chromium (Cr) to tungsten (W). Therefore, gold has a tendency to take a tenth 4d electron from somewhere else to stabilize that shell. Gold (Au), however, is much larger, and its valence electrons do not enjoy such a large energy advantage upon rearrangement, so they do not. The same is true of silver, with electron configurationĬopper and silver are lower-energy "excited states" of what we might have thought would have been the "ground state" of these atoms. With a full d-shell, the propensity for copper to acquire another is low. You will recall that it is energetically favorable for copper to fill its 3d shell (10 electrons) with one of its 4s electrons. Transition (d-block) elementsĬopper (Cu) and silver (Ag) are exceptions to the rule of filling electrons by lowest-energy level first. It has a middle-of-the-road electronegativity, reflecting its high propensity for forming covalent bonds. the H-atom is a bare proton relatively unsheilded by its single 1s electron. More electron shells are added moving down a group, so the outermost electron becomes increasingly distance from the nucleus.It's worth studying some of the details of the periodic table above. There are more protons in atoms moving down a group (greater positive charge), yet the effect is to pull in the electron shells, making them smaller and screening outer electrons from the attractive force of the nucleus. This is because the principal quantum number of the outermost electron increases moving down a group. Ionization decreases moving top to bottom down an element group (column).In general, the size of an atom will decrease as you move from left to the right of a certain period. The noble gas has a filled valence shell, so it resists electron removal. An atom gets larger as the number of electronic shells increase therefore the radius of atoms increases as you go down a certain group in the periodic table of elements. Ionization is at its minimum value for the alkali metal on the left side of the table and a maximum for the noble gas on the far right side of a period. This is because the atomic radius generally decreases moving across a period, so there is a greater effective attraction between the negatively charged electrons and positively-charged nucleus. Ionization energy generally increases moving from left to right across an element period (row).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
